For people diagnosed with lung cancer and their families, understanding the illness and its treatments enough to participate in healthcare decisions can be challenging.
Patients may not understand the benefits of personalized treatment or how to obtain it, whether that involves ensuring that tumor tissue undergoes biomarker testing, choosing among standard treatments, or seeking novel therapies through clinical trials. While experiencing the distress of a diagnosis, patients can find it difficult to undertake the research needed to meet those milestones, or even to determine where to start.
Having recognized an unfulfilled need for tailored information and guidance in this population, the GO2 Foundation for Lung Cancer created the LungMATCH program in 2017 to provide free, unlimited treatment navigation to patients and caregivers at any point along the lung cancer treatment journey. The program encourages biomarker testing and interprets the results, explains a patient’s disease and details about treatment options, and searches nationally for individually relevant clinical trials. It also offers free educational materials such as information sheets, brochures, and a quarterly educational newsletter.
Based on the tenet that patient education and navigation can lead to better utilization of precision medicine, the program is demonstrating success, according to findings shared by a team of researchers from the GO2 Foundation during the IASLC’s 2021 World Conference on Lung Cancer (Abstract OA10.03).
Daniel Saez, MSc, GO2’s treatment and trials navigator, said that recent study results position LungMATCH as a model of best practices for empowering patients to receive personalized medicine and participate in clinical trials.
Tracking Interventions
At the heart of LungMATCH are one-on-one conversations between trained navigators and patients and/or their caregivers, Mr. Saez said.
“We find that the earlier in the treatment journey that patients talk to a LungMATCH navigator, the more repeat conversations they have and the more satisfaction they have from the program,” he said. “Additionally, you can think of LungMATCH as a support for treatment teams, enabling patient interactions to become more efficient at each visit.”
Using records of phone or email contacts made through the program from 2018 through 2020, Mr. Saez and colleagues collected and evaluated data across several categories: patient-reported biomarker testing; history of all treatments received; and clinical trial participation.
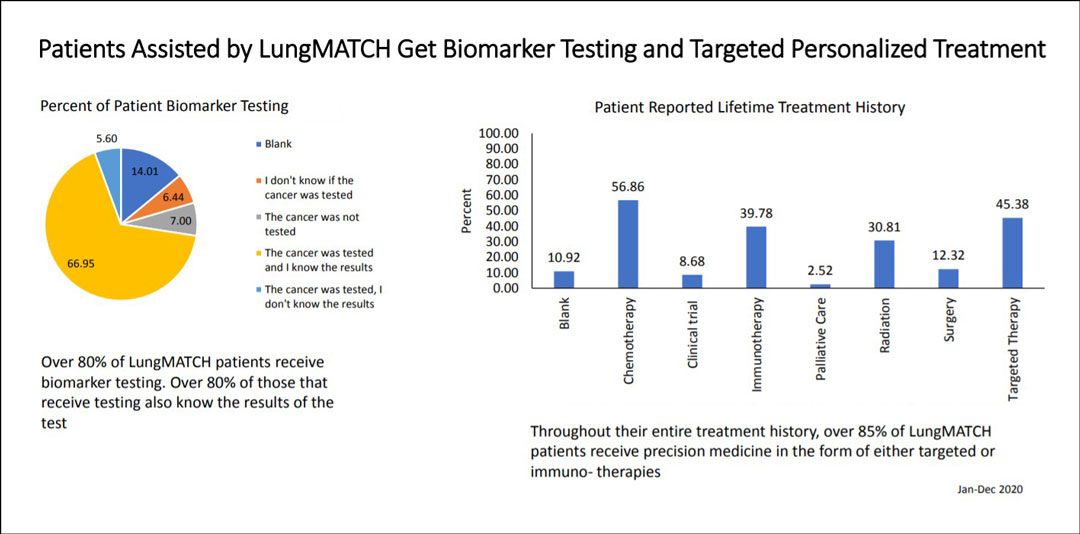
They found that, as the study matured, patients and caregivers reported an increased incidence of biomarker testing and of treatment with targeted drugs and/or immunotherapies.
“Because LungMATCH aims to educate and empower patients, one of the important metrics we look at is our rate of biomarker testing and how that is reflected in the treatment history that patients receive,” Mr. Saez said.
Clinical trial accrual rates, which increased each year of the study, were found to be higher than the national average. Despite the COVID-19 pandemic, recent participation in the program has been above the 2018 baseline of 267 participants. According to the most recent data, there were 415 participants in 2019 and 357 participants in 2020.
Measuring Success
Participating patients who had both received biomarker testing and knew the results totaled 46% in 2018, 64% in 2019, and 67% in 2020. The use of immunotherapy in the study population was 34% in 2018, 41% in 2019, and 40% in 2020. Strikingly, although 13% of participants reported in 2018 that they had ever received a targeted therapy, that segment grew to 38% in 2019 and 45% in 2020. In 2020, more than 85% of participating patients reported having received some form of precision medicine during their treatment history, much higher than the national average of approximately 5%,1 Mr. Saez said.
“We understand that this is [largely] due to the advent of targeted therapies between 2018 and 2020, but it’s still indicative of patients who not only are empowered and educated about their options, but then go on to apply them in their treatment,” he said.
Mr. Saez pointed to an additional measure of success: In 2020, two-thirds of the patients who sought clinical-trial matching through LungMATCH but had never discussed trials with their treatment teams went on to have those discussions. This is about 25% of all patients who used the LungMATCH clinical trial-matching services. Furthermore, he said, 14.3% of those who received navigation through the program that year enrolled in trials, about double the national average. Clinical trial participation among study participants was 11.5% in 2018 and 13.6% in 2019.
Discussant Joshua Sabari, MD, of the Perlmutter Cancer Center at NYU Langone, said that he frequently connects people with LungMATCH through his clinical practice because it fosters “a very savvy patient population” likely to experience improved health outcomes as a result of better utilization of precision medicine.
Mr. Saez said that updates are in store for 2022 that will expand the scope of information gathered about patients and create partnerships designed to help LungMATCH reach more participants.
The IASLC proudly partners with the GO2 Foundation for Lung Cancer on myriad programs and research efforts, including the new Caregiver Survey, which will be released in early February 2022. .
References
- 1. Unger JM, Cook E, Tai E, Bleyer A. Role of Clinical Trial Participation in Cancer Research: Barriers, Evidence, and Strategies. Am Soc Clin Oncol Educ Book. 2016;35:185–198.