Findings from a phase I dose-escalation trial of obrixtamig in combination with ezabenlimab took center stage during the 2026 European Lung Cancer Congress (ELCC) in March in Copenhagen, Denmark. Early data appear promising for patients with small cell lung cancer (SCLC) or other neuroendocrine carcinomas (NECs) expressing delta-like ligand 3 (DLL3).

Julien Mazières, MD, PhD, presented the results during the first proffered paper session of the congress.
“I would first like to thank the scientific committee for selecting this abstract,” said Dr. Mazières, Toulouse University Hospital, France. “It’s not very often that we see a phase I trial in SCLC being presented, so thank you for this.”
Dr. Mazières outlined the mechanism of action of obrixtamig and the rationale behind targeting DLL3, which is frequently overexpressed on the surface of NECs—including SCLC. This overexpression is associated with tumor growth, thus making DLL3 a promising therapeutic target.
Obrixtamig is a novel, IgG-like DLL3-targeted T-cell engager. It simultaneously binds to DLL3 on cancer cells and CD3 on T-cells, leading to the formation of a major histocompatibility complex-independent cytolytic synapse, T-cell activation, and the redirection of T-cells toward DLL3-expressing cancer cells.
In preclinical SCLC models, obrixtamig has been shown to upregulate PD-1 and PD-L1, suggesting that combining obrixtamig with the PD-1 inhibitor ezabenlimab may mitigate this adaptive immune checkpoint and potentially enhance antitumor activity. Additionally, obrixtamig has already demonstrated promising efficacy as monotherapy in patients with DLL3-positive tumors.
Study Design
In the ongoing phase I trial, ezabenlimab was administered as a fixed dose of 240 mg every 3 weeks. Obrixtamig was administered weekly in 3-week cycles at escalating doses of 30, 90, 270, 720, and 1,080 µg/kg, respectively. Patients were treated until disease progression, unacceptable toxicity, consent withdrawal, or any other reasons necessitating discontinuation.
The primary endpoint was dose-limiting toxicity (DLT) during the maximum tolerated dose (MTD) evaluation period (cycle 1 plus a 1-week target dose). Other endpoints included objective response rate (ORR), disease control rate (DCR), duration of response (DoR), and progression-free survival (PFS).
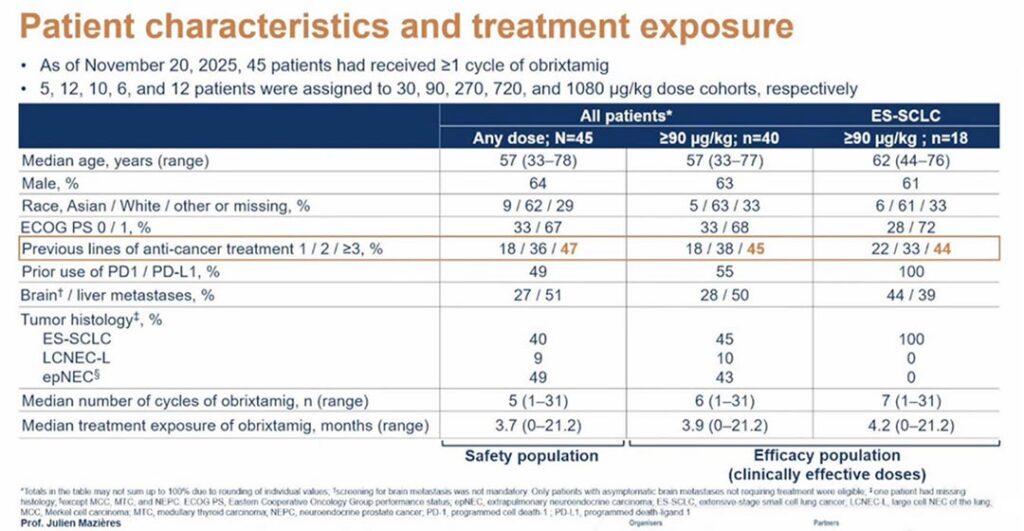
As of November 20, 2025, 45 patients had received at least one cycle of obrixtamig; 40 patients had received a clinically effective dose (defined as 90 µg/kg or more), including 18 with extensive-stage (ES) SCLC.
Most patients had been previously treated, with nearly half having received three or more prior lines of therapy.
Safety Profile
The combination of obrixtamig and ezabenlimab demonstrated a fairly manageable safety profile, Dr. Mazières said.
Treatment-related adverse events (TRAEs) occurred in nearly all patients (98%), with grade 3 or higher TRAEs reported in just over half (51%). Overall, TRAEs leading to dose modifications were minimal:
- Dose reduction of obrixtamig: 1 patient (2%)
- Dose delay of obrixtamig: 5 patients (11%)
- Discontinuation of obrixtamig: 6 patients (13%)
Three DLTs occurred during the step-up dosage phase: one case of grade 2 interstitial lung disease (ILD) and two cases of grade 3 and 4 immune effector cell-associated neurotoxicity syndrome (ICANS). No DLTs were observed at the target dose. Importantly, the MTD was not reached in this trial.
“As expected, the most frequent [adverse events] were cytokine release syndrome (CRS), dysgeusia, fatigue, and decreased lymphocytes,” Dr. Mazières said.
He noted that a significant driver of the frequency of grade 3 or higher events was a decreased lymphocyte count. Additionally, reported CRS events were mainly grade 1 (60%) and grade 2 (18%), with no grade 3 or higher events observed.
Efficacy Outcomes
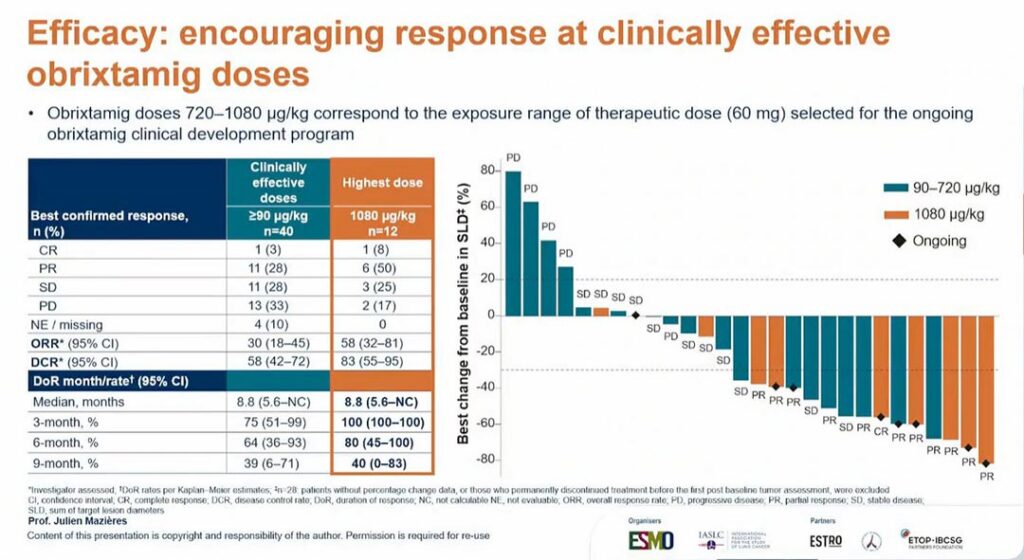
Dr. Mazières reported encouraging responses among patients who received the clinically effective dose or higher.
“If we focus on doses of 90 µg/kg or more, the ORR was 30%, and the DCR was 58%,” Dr. Mazières said. “If we focus on the highest dose, 1,080 µg/kg, you can see that the ORR reached 58% and the DCR was 83%.”
At 6 months, 80% of patients treated with the highest dose remained on treatment. Patients treated with the highest dose also demonstrated promising PFS rates:
- Median PFS: 10 months
- 3-month PFS: 83%
- 6-month PFS: 75%
- 9-month PFS: 55%
Additionally, encouraging efficacy was observed among the 18 patients with ES-SCLC at clinically effective obrixtamig doses.
“Focusing on patients with ES-SCLC, the ORR was 33%, and the DCR was 67%. More than half (56%) of these patients were under treatment after 6 months,” Dr. Mazières said. “The median PFS in patients with ES-SCLC was 5.7 months, and the PFS rate was 48% at 6 months.”
Future Directions
Overall, obrixtamig and ezabenlimab demonstrated a manageable safety profile in heavily pretreated patients with DLL3‑positive ES-SCLC and other NECs, with no additional or unexpected toxicities observed. While CRS was frequently reported, most cases were grade 1 or 2, with no grade 3 events reported.
Encouraging efficacy was observed at clinically effective doses. PFS also appeared promising in patients who received the highest dose, with a median PFS of 10 months and a 6-month PFS rate of 75%.
“These data support the combination of obrixtamig with ICIs and warrant further development for the treatment of patients with SCLC,” Dr. Mazières concluded.
He also noted that the DAREON-8 study is currently investigating obrixtamig in combination with atezolizumab and chemotherapy and is advancing toward further development as a potential first-line treatment for ES-SCLC.










