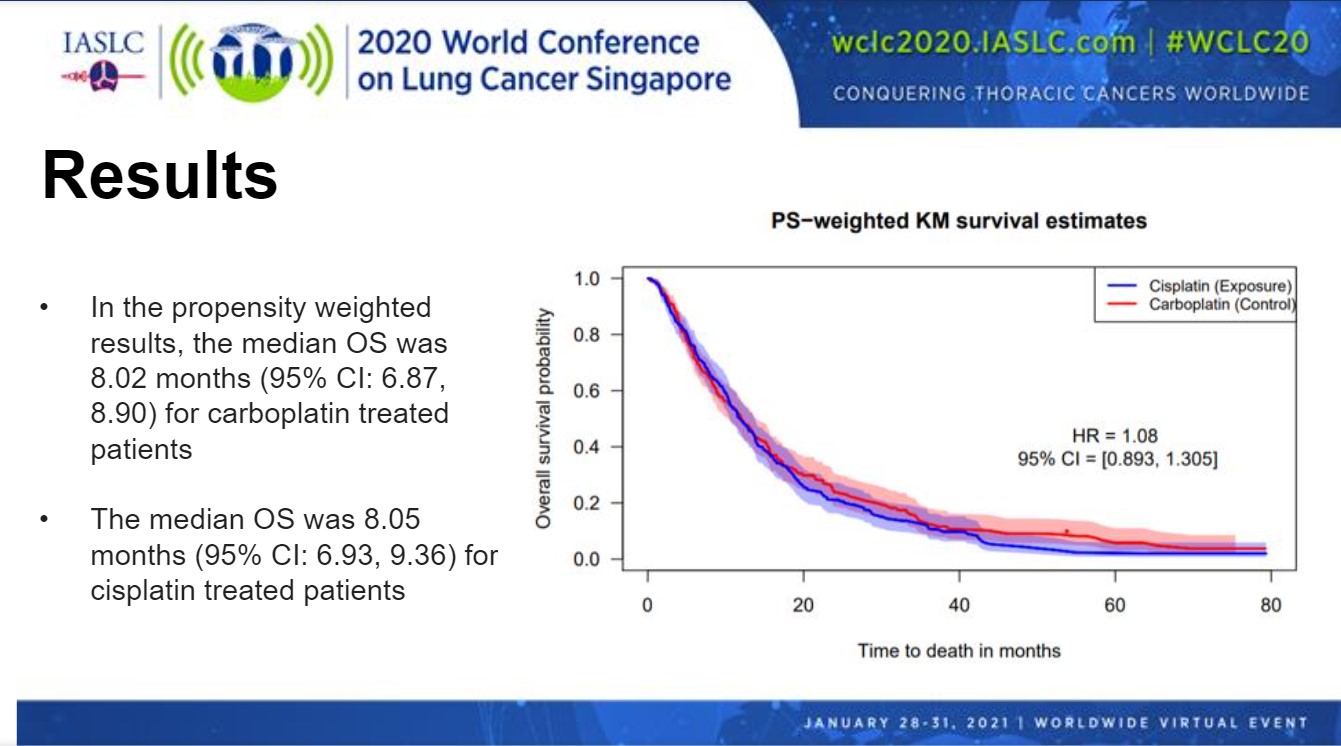
The largest real-world cohort study of individuals with malignant pleural mesothelioma (MPM) found a median survival duration of 8 months regardless of whether patients received first-line treatment with cisplatin-based chemotherapy or carboplatin-based chemotherapy (OA09.05). This bodes well for patients with MPM, who are often elderly, frail, and unable to tolerate cisplatin.
“For MPM patients who are treated with first-line platinum combination chemotherapy in the real-world setting, we found that carboplatin had equivalent performance as compared to cisplatin with respect to overall survival (OS). Thus, we feel that carboplatin is an effective, and potentially less toxic, alternative therapy and can be utilized in the frontline setting in MPM patients,” concluded Kathleen Kerrigan, DO, of the Huntsman Cancer Institute at the University of Utah, who presented the study results.
Patients with MPM face very poor survival outcomes, typically on the order of 12 to 18 months, underscoring the importance of maximizing the precious little time available to these patients. Cisplatin is the preferred platinum chemotherapy backbone endorsed by national guidelines and is the only agent approved for use in MPM, in combination with pemetrexed, given its ability to extend patient survival. Unfortunately, most patients with MPM are ineligible to receive cisplatin based on their comorbidities or poor Eastern Cooperative Oncology Group (ECOG) performance status, prompting many oncologists to prescribe carboplatin instead owing to its better tolerability.
But is carboplatin as effective as cisplatin in MPM?
To address this question, Dr. Kerrigan and colleagues drew from electronic health records in the US Flatiron Health database. Data for 787 patients with MPM who initiated platinum-based therapy between January 2011 through July 2019 were included.
The characteristics of the population reflected those typically seen among MPM patients. For example, 68% had asbestos exposure, 67% had a history of smoking, and 59% had epithelioid histology.
More patients within the cohort received first-line treatment with carboplatin (59%) as opposed to cisplatin (41%). As expected, carboplatin-treated patients were older (median age: 77 vs. 70 years) and had a worse ECOG performance status score (eg, ≥ 2: 11.4% vs. 6.2%) when compared with their cisplatin-treated counterparts.
As Dr. Kerrigan explained, propensity weighting was first performed to balance out all potential confounders, including age; asbestos exposure; smoking history; region; practice type (academic/community); histology; PD-L1 status; patient medical comorbidities, including neuropathy, heart disease, kidney disease, and otopathology; height; weight; ECOG performance status; and serum creatine. Thereafter, OS was analyzed according to the choice of platinum chemotherapy.
“After applying propensity-score weighted analyses, we believe that this essentially removed the confounding risk of baseline features, and thus we feel confident in our conclusion that confounders would not necessarily impact the OS effect of cisplatin,” Dr. Kerrigan stated.
The propensity-weighted results showed that median OS reached 8.02 months for patients treated with a carboplatin combination and 8.05 months for patients treated with a cisplatin combination in the first-line setting (Figure). The hazard ratio (HR) for OS observed with cisplatin relative to carboplatin was 1.08 (95% CI: 0.893-1.305), which was not statistically significant. Based on the confidence interval data, cisplatin-based therapy provides at most an 11% reduction in the risk for death and up to a 34% increase in the risk for death relative to carboplatin-based therapy.
“We feel like our results do provide added context when choosing between the two agents,” Dr. Kerrigan said. “Carboplatin is safe, it’s effective, it’s tolerable, and it has a similar PFS and OS. Thus, we don’t see any obvious reason to choose cisplatin over carboplatin in the real-world clinic setting.”
“A limitation of our study, of course, is that this is a retrospective observational study. Thus, our results may be impacted by unmeasured confounders and missing data, particularly for some elements in this analysis,” Dr. Kerrigan remarked.
Despite these potential limitations, Thomas John, MBBS, PhD, of the Peter MacCallum Cancer Centre in Melbourne, Australia, indicated that Dr. Kerrigan’s findings corroborate those of an older study conducted in 2008 that showed similar survival outcomes in MPM with use of either cisplatin- or carboplatin-based treatment.1 This analysis of 1702 chemotherapy-naïve patients who received platinum-based therapy under an expanded access program reported 1-year survival rates of 63.1% with cisplatin/pemetrexed and 64.0% with carboplatin/pemetrexed.
“I think we can conclude, even though these are not randomized studies, that cisplatin does appear to behave very similarly to carboplatin,” Dr. John concluded. “The next question is: where does immunotherapy sit in this?”
Dr. Kerrigan raised the same point. “It remains to be seen how this presentation will affect the treatment paradigm of first-line MPM,” she stated. This is because recent data from CheckMate 743 showed that the combination of nivolumab plus ipilimumab in the first-line setting improves median OS by approximately 4 months compared with chemotherapy (18.1 vs. 14.1 months; HR 0.74; 95% CI 0.61-0.89; p = 0.002).2
“The data is great and exciting, and we look forward to seeing how real-world patients also respond,” Dr. Kerrigan remarked.
This session had a real-time Q&A that provided attendees with the opportunity to ask questions of the session participants. The Q&As are included in the On-Demand recordings, available through the virtual platform. Registration is ongoing for the next 90 days at wclc2020.iaslc.org.
References
- Santoro A, O’Brien ME, Stahel RA, et al. Pemetrexed plus cisplatin or pemetrexed plus carboplatin for chemonaïve patients with malignant pleural mesothelioma: results of the International Expanded Access Program. J Thorac Oncol. 2008;3(7):756-763.
- Baas P, Scherpereel A, Nowak AK, et al. First-line nivolumab + ipilimumab vs chemotherapy in unresectable malignant pleural mesothelioma: CheckMate 743. Presented at the IASLC WCLC 2020 Virtual Presidential Symposium, August 8, 2020. Available at: https://wclc2020.iaslc.org/wp-content/uploads/2020/08/Abstracts-Virtual…;