-
Interim NADIM ADJUVANT Data Suggest Adjuvant Chemo-IO May Reduce Recurrence Risk in Patients Following Complete Resection
-
Final FLAURA2 OS Data Show Osimertinib Plus Chemo Offers Benefit Compared with Monotherapy
-
Potential Impact of GLP1-RAs on TKI-induced Weight Gain in Patients with NSCLC
-
No “Known” Risk Factors: The Health Consequences of Radiation Therapy
-
Discussion Covered Updates, Strategies, and Controversies in Lung Cancer Staging
-
No PFS, OS Benefit Seen in Final Results from PACIFIC-2
-
WCLC Attendees Hear Preview of Proposed Changes for the 9th Edition of the TNM Staging Classification for Thoracic Cancers
-
Treatment of Immunotherapy-Related Dermatologic Toxicities: An Interview with Dr. Mario Lacouture
-
A Holistic Approach to Patient Care
Evolving Standards of Care
-
As Treatment Options in Lung Cancer Have Expanded, so Have Costs
A real-world look at diagnostic-therapeutic pathways explores how they may help provide optimal, innovative care without ballooning costs.
-
Small, High PLCG2-Expressing Intratumoral Cells May Drive Metastasis
In the search to understand the mechanisms of metastasis, researchers have expanded the spectrum of SCLC clinical specimens amenable for single-cell resolution analysis.
-
COAST Data Support Further Research of Immunotherapy Combinations
Trial is the first to show improved efficacy and a manageable safety profile after cCRT in unresectable locally advanced stage III NSCLC regardless of PD-L1 status.
-
TIL Therapy Endures for Carefully Selected Patients: A Q&A with Dr. Benjamin C. Creelan
Dr. Benjamin C. Creelan shares his passion for cell therapy research and insights into current data.
-
Early Palliative Care: A European Perspective
The benefits of early palliative care have been proven. Could negative connotations of palliative care be impeding its adoption?
-
Tumor Growth Rate as a Guide for Precision Lung Cancer Therapy
The speed at which a tumor progresses may prove to be a useful tool in therapeutic decision-making.
-
Patient Perspective: The Promise of Personalized Medicine
Growing treatment options for patients with late-stage lung cancer bring hope—and challenges.
-
Cancer Misinformation: Its Impact on Patients and Mitigation Strategies
Inaccurate health information can have grave consequences, but there are techniques providers can use to combat it.
-
A Look at Veliparib Plus Chemotherapy for First-Line Treatment of Advanced NSCLC
The strategy likely will not be pursued, however studies of PARP inhibitors plus immune checkpoint inhibitors are already in advanced stages.
-
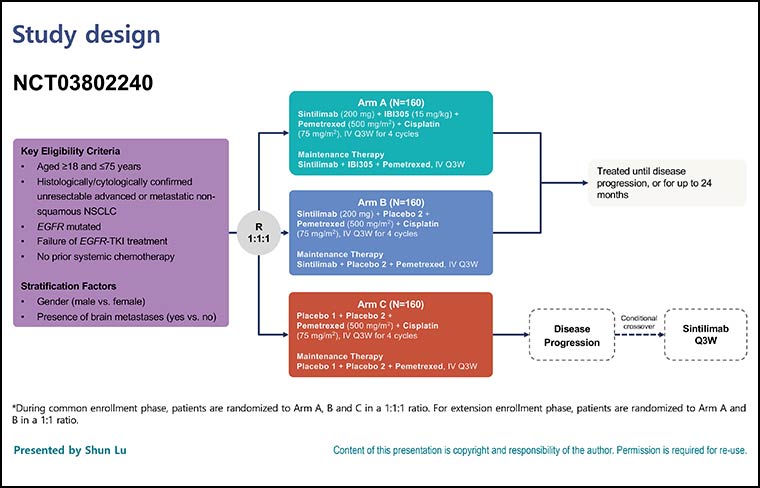
A New Standard of Care in EGFR-Positive NSCLC Unresponsive to TKIs?
Interim analysis from ORIENT-31 demonstrates a four-drug regimen may be a reasonable option for patients whose disease has progressed on EGFR TKIs.