China’s Domestic PD-1/PD-L1 Inhibitors Demanding Global Attention
Leah LawrenceThe growth of these domestic drugs should shine a light on the lack of diversity in many international clinical trials in immunotherapy. Read more
Biomarkers May Illuminate the Path Forward for PARP Inhibitors in SCLC
Marcelo V. Negrao+more
Despite U.S. Food and Drug Administration (FDA) approvals for the addition of anti–programmed death-ligand 1 (PD-L1) therapies to a first-line combination of etoposide plus a platinum-based agent for extensive-stage small […] Read more
NCI Launches the Cancer Moonshot SM Biobank to Accelerate Research in Cancer Treatment
Helen M. Moore, PhDThe U.S. National Cancer Institute (NCI) Cancer Moonshot Biobank launched in September 2020, with the aim of accelerating cancer research by creating a resource of well-annotated, longitudinal cancer biospecimens from […] Read more
Voice from Asia: Combination of Immune Checkpoint Inhibitors With Chemotherapy for Advanced Non-squamous NSCLC
Weigang Xiu, MD, PhD+more
The immune checkpoint inhibitors (ICIs) monotherapy has resulted in significant improvements in PFS and OS in advanced NSCLC when compared with chemotherapy alone, and it has become the standard of […] Read more
ASCO and Ontario Health Publish Joint Guideline Update on Systemic Therapy for Stage IV NSCLC
Dr. Joan SchillerIn February 2021, the American Society of Clinical Oncology (ASCO) and Ontario Health (Cancer Care Ontario) published a joint guideline update to the 2017 ASCO guidelines regarding systemic therapy for […] Read more
Immunotherapy Use in Patients With Lung Cancer and Comorbidities
Mitchell von Itzstein+more
Because nearly one-half of patients with advanced cancer are eligible for immune checkpoint inhibitor (ICI)–based treatment, these therapies are now in widespread clinical use.1 ICIs—whether as monotherapy, in checkpoint inhibitor […] Read more
Increasing Understanding about the Role Biosimilar Agents Play in Everyday Practice: A Q&A with Dr. Gary Lyman (Part 2 of 2)
Gary Lyman, MD, MPHIn September 2017, the US Food and Drug Administration (FDA) approved bevacizumab-awwb for the treatment of NSCLC, with the same indications as the tradename. Although bevacizumab-awwb is the only biosimilar […] Read more
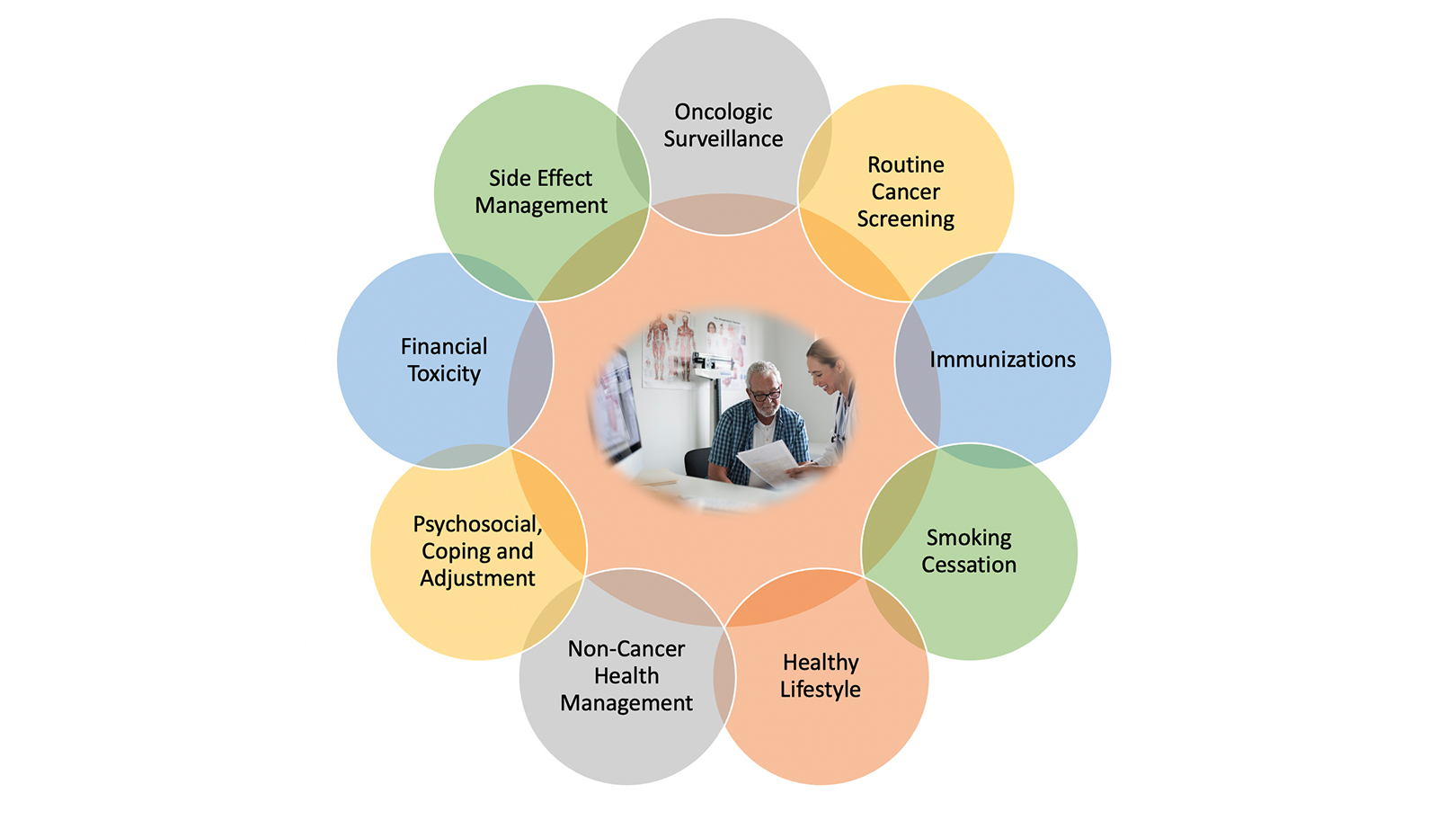
Ensuring Quality of Life During Lung Cancer Survivorship Requires a Multidisciplinary Team
Carolyn Vachani, RN, MSN+more
Lung cancer is one of the most common cancers worldwide, with more than 2 million people diagnosed each year.1 Advances in early detection and treatment have resulted in more people […] Read more
COVID-19 and Lung Cancer: A Call for Immediate Action
David R. Baldwin, MD, FRCPThe COVID-19 pandemic has had a devastating effect on lung cancer outcomes in the United Kingdom (UK). Fewer people have presented or have been referred for investigation, and those who […] Read more
A Clinical Trial of PD-L1 Inhibitor Durvalumab and PARP Inhibitor Olaparib for Patients With EGFR-Mutated Transformed SCLC
Nobuyuki Takahashi, MD, PhD+more
Discovery of targetable genomic alterations, including mutations of EGFR, have resulted in improved survival for patients with non-small cell lung cancer (NSCLC).1 Although many patients experience robust responses to EGFR-targeted […] Read more